

Physiological studies on Chinese Water Chestnut
Li, M.; Lyall, T.; Kleinhenz, V.; Midmore, D. J., 1998
Access to Asian Foods, 3, 7
Physiological Studies on Chinese Water Chestnut
M Li, T Lyall, V Kleinhenz and D J Midmore
Chinese water chestnut is a crop new to Australia. Within the few years of commercial production a wide range of variation in corm size and sweetness have been observed by producers. Corms harvested in New South Wales (NSW) and Victoria (Vic) tend to contain much higher soluble solids (sugars) than those harvested in Queensland (Qld). Since consumer acceptance depends to some degree on the sweetness of corms, it is important to understand what determines sweetness in corms. Is it due to different genotype, or to maternal corm effects (i.e. of the conditions prior to production), or simply due to differences in environmental factors and cultivation management during production? It has also been noted that Chinese water chestnut harvest, wherever grown to date in Australia, is never earlier than June, and extends to October. This limits the availability of fresh supply to the market. Cold storage to some extent offsets this, but could prove to be uneconomical for the storage of large quantities of corms.
Both the questions of corm sweetness, and of harvest timing, have been addressed by the Central Queensland University Asian vegetables research team. Initial results are presented here.
Corm sweetness
Using molecular tools (random amplified DNA (RAPD)) that essentially indicates whether the DNA from one plant is the same or different to that of another plant, we have been able to analyse leaf and/or corm samples from across Australia, and compare them to others from Taiwan, PR of China and the USA. Results show that materials from Taiwan (var. Shu-Lin) and Hangzhou (PR of China) (var. Da Hong Bao) were very different from other materials. Materials from Coffs Harbour and Georgia (USA) were also somewhat dissimilar to the rest of the samples. The remainder of those collected in Australia were essentially the same (based upon the degree of precision of our methods). That most material in Australia is genetically similar suggests that maternal effects and/or the current growing conditions are more important than "variety" in controlling sweetness in corms. An experiment is underway in which three "varieties" are grown in Vic, NSW and Qld. The harvests will be split in three ways to be regrown in the next season at each site. This way, we can again determine whether "variety" or previous/current growing environment is more important in growing corm sweetness.
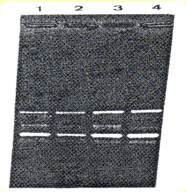
RAPD profile of stem and corm material for one - "variety" showing the apparent identical genetic makeup, as expected, for different parts of the same plant. In plants with different genetic makeup, the positions of the bands would not coincide
Harvest timing
We set up a series of trials in which we artificially altered the photoperiod (i.e., the daily length of the light period) by either turning on lights above plants during the evening and into the night to make them believe they were growing under daylengths longer than the natural, or by transferring plants into dark, light-proof compartments before the end of the natural daylength to make them believe they were growing in daylengths shorter than the natural. The use of lights to "extend" the daylength is common to some cutflower industries. This induces plants that normally flower in the summer to flower in the winter.
As for many plant species, Chinese water chestnut exhibits a response to photoperiod. Photoperiods ranging from 8 to 24 h did not significantly affect plant dry matter. Dry matter partitioning to stems and roots did not differ much for photoperiods ranging 11 to 24 h. Under 8 h photoperiod however, dry matter allocated to stems was greatly reduced with the balance found in newly-formed corms. The effect on corm number is shown in Figure 1.
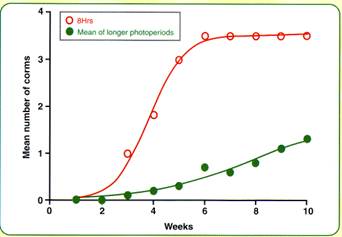
Figure 1. Effect of short and long photoperiods of water chestnut corms produced
Corm formation and growth was inhibited by long photoperiods (16, 20, 24 h) and promoted by short photoperiods. When corms were formed under long day treatments, they were presumably the result of the inducing effects of short natural photoperiods before the imposition of treatments. Once induction was effected, it was not reversed by long photoperiods. However, under prolonged long photoperiods, no more corms were formed and some resorption of carbohydrate from small immature (< 1.0 cm diameter) was indicated.
Constant short photoperiods (£ 13 h), shortening photoperiods from 13 h to 11 h, or short natural photoperiods (11.0-13.7 h) brought about corm formation. The changing pattern of natural photoperiods, whether shortening or lengthening, seemed less important for corm formation than the length of day being below a critical maximum for corm formation. However, this critical value, which is probably between 13-16 h is still not known. The determination of critical photoperiod will be subject to the modifying effects as temperature and irradiance level.
Among photoperiods that were shorter than the critical value, the shortest photoperiod tended to more strongly induce corm formation. Under 8 h photoperiod, a few corms were observed to be directly formed on mother corms, ie without rhizomes. This has also been observed in potato under highly inductive conditions. The inducing effects of the short photoperiod treatments (8-13 h) were closely related to exposure time. Shorter photoperiods required fewer inductive cycles for corm formation than did longer photoperiods. The longer the short day treatment, the stronger the inductive effect.
Based upon these results, we can now manipulate photoperiod artificially to induce Chinese water chestnut to form corms almost at will.

School of Biological and Environmental Sciences
Central Queensland University
Rockhampton QLD 4702
Tel: (07) 4930 9770
Fax: (07) 4930 9255